

|
|
|
|
Independent Research and Development, global source and supply for the domestic heating stove industry. Wood is most of the world's renewable energy, it is more efficient and pays back investment quicker than anything. We're developing today's ultra-clean, controllable stoves... 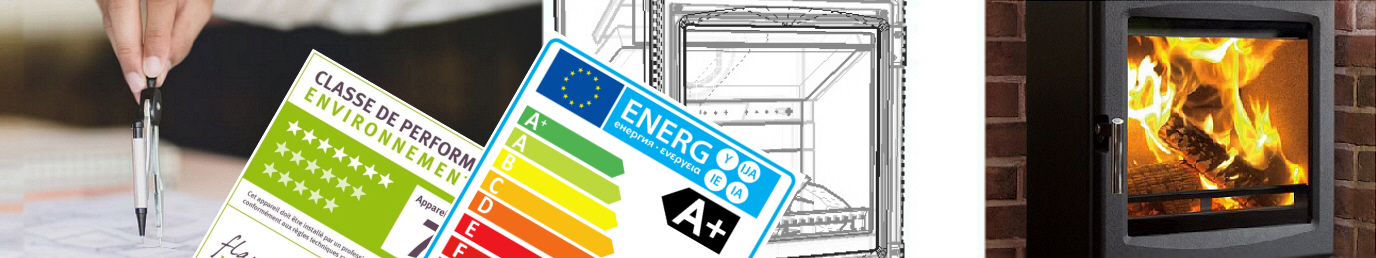 WE DESIGN Domestic heating, cooking stoves and boilers to today's ultra-low emission, high efficiency Standards WE TEST With our academic partners we have an ability - unique in the world - to measure gravimetric emissions in real time, as well as CO, CO2, HxCy, NOx. This instant test ability, and our close relationship with assay laboratories, means we develop better products much, much faster.  WE SOURCE OEM components from iron castings, fabrications and ceramics to windows and seals  WE SUPPLY some of the best-known names in Europe and North America BrightStove is a consortium of design, development and testing companies with reliable partnerships across the world. We design and develop domestic solid fuel stoves, boilers, cookers, furnaces and heaters to meet all new and existing Standards. Our intimate connections with Standards Agencies mean that we're always ahead of the game. We supply castings, fabrications, windows, seals, lining components and complete stoves. Vitreous enamel, Cr/Fe casting, silk screening, plating, polishing, siloxane coatings, ceramics, precision turning... all with low cost tooling and rapid turnarounds. We supply the biggest stove manufacturer in North America, and we supply the biggest stove manufacturer in Europe. But we also supply a number of tiny one-person businesses. And we can supply you. |
|
MORE FROM BrightStove... Home Condensing Stove Images The Clean Living Fire Email: info@brightstove.com COPYRIGHT and ALL RIGHTS RESERVED: © BrightStove Consortium, 19/11/2020 BUILT WITH WHIMBERRY matrixstats |